







《《氮及其化合物 硝酸 酸雨及防治》人教版高中化学必修二教学PPT课件》是由用户上传到老师板报网,本为文库资料,大小为600.69 KB,总共有35页,格式为pptx。授权方式为VIP用户下载,成为老师板报网VIP用户马上下载此课件。文件完整,下载后可编辑修改。
- 文库资料
- 35页
- 600.69 KB
- VIP模板
- pptx
- 数字产品不支持退货
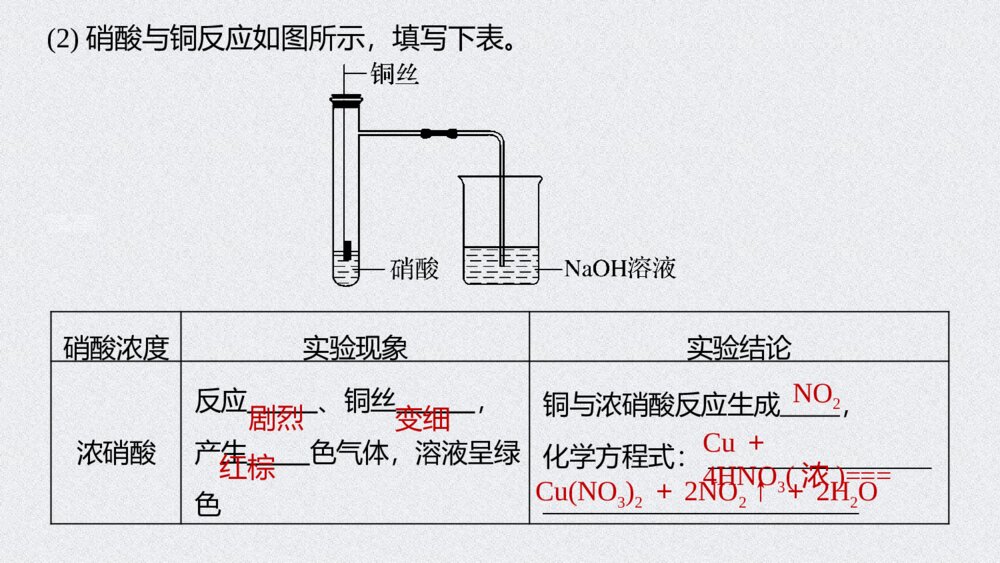
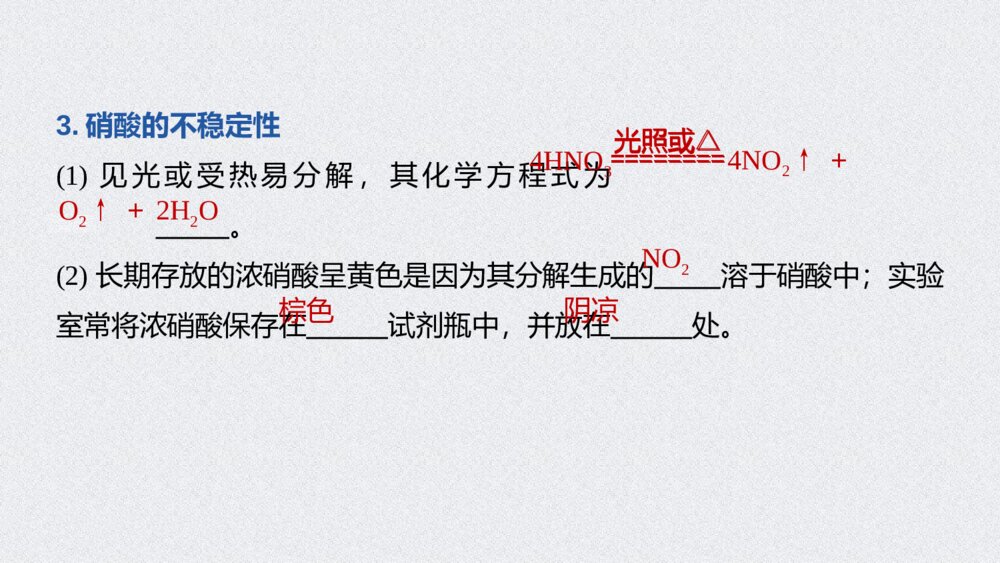
第3课时 硝酸 酸雨及防治第五章 第二节 氮及其化合物1.能从物质类别、氮元素化合价的角度认识硝酸的性质及含氮物质间的相互转化,促进“证据推理与模型认知”化学核心素养的发展。2.认识二氧化硫、氮氧化物为大气污染物,了解酸雨的形成、危害及其防治,增强环保意识和社会责任感。核心素养发展目标二、酸雨及防治内容索引NEIRONGSUOYIN一、硝酸的性质硝酸的性质XIAOSUANDEXINGZHI01PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/1.硝酸的物理性质纯硝酸为无色、有气味的液体,沸点低,易,在空气中呈“白雾”状,质量分数为95%以上的浓硝酸称为“硝酸”。2.硝酸的强氧化性(1)浓硝酸中的+5价氮元素具有很强的得电子能力。浓硝酸和稀硝酸都具有很强的氧化性,且浓度越大氧化性越强,还原剂一般被其氧化为最高价态。知识梳理刺激性挥发发烟PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(2)硝酸与铜反应如图所示,填写下表。硝酸浓度实验现象实验结论浓硝酸反应、铜丝,产生色气体,溶液呈绿色铜与浓硝酸反应生成,化学方程式:_________________________________________剧烈变细红棕NO2Cu+4HNO3(浓)===Cu(NO3)2+2NO2↑+2H2OPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/稀硝酸反应、铜丝,产生气体,溶液呈蓝色铜与稀硝酸反应生成,化学方程式:________________________________________________缓慢变细无色NO3Cu+8HNO3(稀)===3Cu(NO3)2+2NO↑+4H2OPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/根据浓硝酸和稀硝酸的强氧化性书写下列反应的化学方程式:①铁与稀硝酸(HNO3过量):。②碳与浓硝酸共热生成CO2和NO2:。③硫与浓硝酸共热生成H2SO4和NO2:。Fe+4HNO3(稀)===Fe(NO3)3+NO↑+2H2OC+4HNO3(浓)=====△CO2↑+4NO2↑+2H2OS+6HNO3(浓)=====△H2SO4+6NO2↑+2H2OC+4HNO3(浓)=====△CO2↑+4NO2↑+2H2OS+6HNO3(浓)=====△H2SO4+6NO2↑+2H2OPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/思维拓展(1)硝酸与金属反应不能生成氢气。硝酸浓度不同,其还原产物不同,一般情况下,浓硝酸被还原为NO2,稀硝酸被还原为NO。(2)硝酸能与大多数金属(金、铂等除外)反应,将金属氧化为高价态金属的硝酸盐。但在常温下,浓硝酸能使铁、铝钝化。(3)热的浓硝酸可将非金属单质(碳、硫、磷等)氧化为最高价氧化物或最高价含氧酸。(4)硝酸的强氧化性还表现在可以氧化具有还原性的化合物或离子,如SO2、FeO、Fe2+、Br-、I-、S2-、SO等均能被硝酸氧化。2-32-3PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/3.硝酸的不稳定性(1)见光或受热易分解,其化学方程式为。(2)长期存放的浓硝酸呈黄色是因为其分解生成的溶于硝酸中;实验室常将浓硝酸保存在试剂瓶中,并放在处。4HNO3========光照或△4NO2↑+O2↑+2H2ONO2棕色阴凉========光照或△PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(1)硝酸具有腐蚀性和挥发性,使用时要注意防护和通风( )(2)稀硝酸可溶解Fe(OH)2生成Fe(NO3)2和水( )(3)碳、铜与浓硝酸反应时,浓硝酸只表现强氧化性( )(4)浓硝酸、稀硝酸都具有强氧化性,它们都能使铁、铝钝化( )(5)王水是浓硝酸和浓盐酸体积比为1∶3的混合物,其氧化性比浓硝酸强( )判断正误√×××√PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/回答下列问题:(1)如何除去浓硝酸中的“黄色”?提示 在浓硝酸中通入足量的氧气,发生反应4NO2+O2+2H2O===4HNO3,使NO2转化为硝酸,即可除去浓硝酸中的“黄色”。(2)足量的铜与浓硝酸反应,浓硝酸能否完全被还原?其还原产物可能是什么?提示 浓硝酸不可能完全被还原,因生成的硝酸铜溶液中仍有NO;在反应过程中,硝酸的浓度逐渐减小,还原产物可能是NO2和NO的混合物。(3)将铁粉溶于稀硝酸,铁元素的存在形式可能为①________、②________、③__________________。深度思考-3Fe(NO3)3Fe(NO3)2Fe(NO3)3和Fe(NO3)2-3PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/思维感悟认识无机物的主要角度PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/相关链接(1)氧化性酸和酸的氧化性①浓硫酸、硝酸等都是氧化性酸,因为其中的易获得电子,表现出很强的氧化性;而盐酸、稀硫酸、碳酸等的酸根部分不能或不易获得电子,是非氧化性酸。②在水溶液中任何酸都能电离出H+,H+在一定条件下可获得电子,被还原为H2,从这一点来说,酸都具有氧化性,但此时H+表现出的性质是酸性,这与氧化性酸中心元素获得电子表现出的氧化性不同。S+6、N+5S+6、N+5PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(2)“三酸(浓盐酸、浓硫酸、浓硝酸)变稀”问题辨析。①浓盐酸与足量的二氧化锰反应时,浓盐酸浓度逐渐变小,变为稀盐酸时不再与二氧化锰反应。②浓硫酸与足量的铜反应时,浓硫酸浓度逐渐变小,变为稀硫酸时不再与铜反应。③浓硝酸与足量的铜反应时,浓硝酸浓度逐渐变小,变为稀硝酸时,还原产物发生改变,生成NO。解析 浓硝酸在见光或受热时会发生分解反应生成氧气、二氧化氮和水,A项正确;硝酸是氧化性酸,金属与硝酸反应不产生氢气,B项正确;常温下铁和铝在浓硝酸中钝化,因此可用铁和铝制品盛装冷的浓硝酸,C项正确;铜与硝酸反应生成硝酸铜、氮的氧化物和水,不属于置换反应,D项错误。1.下列对于硝酸的认识,不正确的是A.浓硝酸在见光或受热时会发生分解B.金属与硝酸反应不产生氢气C.可用铁和铝制品盛装冷的浓硝酸D.铜与硝酸的反应属于置换反应跟踪强化123√2.0.3mol铜和含1.2molHNO3的浓硝酸,充分反应后,生成NO2的量为A.0.2molB.0.6molC.小于0.6molD.大于0.6mol解析 因铜与浓硝酸反应:Cu+4HNO3(浓)===Cu(NO3)2+2NO2↑+2H2O,随着反应的进行,浓硝酸变稀,稀硝酸与铜反应:3Cu+8HNO3(稀)===3Cu(NO3)2+2NO↑+4H2O,所以放出的NO2小于0.6mol。123√3.氮及其化合物的变化关系如下所示。123解析 氮气和氧气在放电条件下反应生成NO,NO与O2反应生成NO2,氮气与氧气不能一步转化为NO2,D错误。返回则下列说法不正确的是A.路线①②③是工业生产硝酸的主要途径B.路线Ⅰ、Ⅱ、Ⅲ是雷电固氮生成硝酸的主要途径C.上述所有反应都是氧化还原反应D.氮气可在足量的氧气中通过一步反应生成NO2√02酸雨及防治SUANYUJIFANGZHIPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/1.酸雨的概念正常雨水的pH约为5.6,这是因为雨水中溶解了的缘故。pH<5.6的降水称为酸雨。2.酸雨的形成(1)大气中的、及它们反应后的生成物溶于而形成酸雨。知识梳理二氧化碳二氧化硫氮氧化物雨水PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(2)硫酸型酸雨的形成过程写出图中标号所示的化学方程式:①;②;③;④。2SO2+O2粉尘等催化剂2SO3SO2+H2OH2SO3SO3+H2O===H2SO42H2SO3+O2===2H2SO42SO2+O2粉尘等催化剂2SO3SO2+H2OH2SO3PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(3)硝酸型酸雨的形成过程汽车尾气―→NO――→O2①NO2――――→雨水②HNO3写出图中标号所示的化学方程式:①;②。2NO+O2===2NO23NO2+H2O===2HNO3+NO汽车尾气―→NO――→O2①NO2――――→雨水②HNO3PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/3.酸雨的危害(1)直接农作物,破坏森林和草原,使土壤、湖泊。(2)加速建筑物、桥梁、工业设备、运输工具和电缆的。(3)导致地下水中重金属元素含量增加,污染水源,危害人体健康。4.酸雨的防治(1)消除,改变能源结构,开发利用清洁能源,如太阳能、风能、氢能等。(2)对的排放加以控制,如燃料脱硫处理,废气中二氧化硫回收利用、控制汽车尾气排放等。(3)健全法律法规,严格规定污染物的排放标准,提高环境保护意识。损伤酸化腐蚀污染源酸性物质1.现有雨水样品1份,每隔一段时间测定该雨水样品的pH,所得数据如下:理解应用测试时间/h01234雨水的pH4.734.634.564.554.55(1)分析数据,回答下列问题:①雨水样品的pH变化的原因是________________________________________(用化学方程式表示)。SO2+H2OH2SO3,2H2SO3+O2===2H2SO4解析 酸雨放置时pH变小,其原因是雨水中的H2SO3被空气中氧气氧化所致:SO2+H2OH2SO3,2H2SO3+O2===2H2SO4。SO2+H2OH2SO3,②如果将刚取样的上述雨水和自来水相混合,pH将变___,原因是__________________________________________________________(用化学方程式表示)。小Cl2+2H2O+SO2===H2SO4+2HCl或H2SO3+HClO===H2SO4+HCl解析 雨水和自来水混合,应考虑SO2与Cl2(或H2SO3与HClO)的反应:SO2+2H2O+Cl2===H2SO4+2HCl(或H2SO3+HClO===H2SO4+HCl)。因此雨水与自来水混合后溶液pH将变小。(2)下列措施中,可减少酸雨产生的途径的是____(填字母)。①少用煤作燃料 ②把工厂烟囱造高 ③燃料脱硫④在已酸化的土壤中加石灰 ⑤开发新能源A.①②③B.②③④⑤C.①③⑤D.①③④⑤解析 减少酸雨的产生需减少SO2的排放,将煤转化为清洁能源(如煤的气化和液化),燃料脱硫、固硫,开发不含硫的新能源(如氢能、核能等),都是有力措施。因此①③⑤正确。CPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/2.回答下列问题(1)我国城市环境中常见的大气污染物主要有哪些?提示 SO2、NO2、CO、烟尘等。(2)根据所学知识,你知道采取什么办法能解决下列问题吗?①治理已酸化的湖泊和土壤。提示 对于已酸化的湖泊和土壤,可以加入生石灰进行改良。②保护暴露在大气中的建筑。提示 对于暴露在大气中的建筑可以通过粉刷油漆进行保护。PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(3)某有色金属冶炼厂排放的废气中含SO2,先用石灰浆吸收SO2,然后利用空气中的氧气将产物继续氧化成石膏(CaSO4·2H2O)。写出有关反应的两个化学方程式。提示 SO2+Ca(OH)2===CaSO3+H2O,2CaSO3+O2+4H2O===2CaSO4·2H2OPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/相关链接常见的环境污染环境污染形成原因主要危害温室效应大气中CO2含量不断增加全球变暖,冰川融化酸雨SO2和氮氧化物的排放土壤酸化,腐蚀建筑物光化学烟雾氮氧化物和碳氢化合物的排放危害人体健康和植物生长臭氧空洞氮氧化物和氟氯代烃的排放地球上的生物受太阳紫外线的伤害加剧PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/赤潮和水华含磷洗衣粉的大量使用及废水的任意排放使藻类过度繁殖,水质恶化,发生在海水中为赤潮,淡水中为水华白色污染聚乙烯塑料的大量使用、任意丢弃破坏土壤结构和生态环境1.下列有关酸雨的说法正确的是A.凡是呈酸性的雨水都称为酸雨B.酸雨的形成只与SO2的排放有关C.煤的脱硫可以在一定程度上防止酸雨的形成D.为了不形成酸雨,应停止使用一切化石燃料跟踪强化123√解析 A选项,正常雨水中由于溶解了二氧化碳,也呈酸性,pH<5.6的降水称为酸雨;B选项,酸雨分为硝酸型酸雨和硫酸型酸雨,硝酸型酸雨的形成与氮氧化物的排放有关;C选项,利用石灰石等物质,使燃煤中的含硫物质最终转化为硫酸钙,达到脱硫的目的,减少SO2的排放,在一定程度上可防止酸雨的形成;D选项,化石燃料是现代社会的主要能源,不可能一点都不使用,应推广使用脱硫技术,大力开发清洁环保的新能源,减少酸雨的形成。1231232.现有以下几种措施:①对燃烧煤时产生的尾气进行除硫处理;②少用原煤作燃料;③燃煤时鼓入足量空气;④开发清洁能源。其中能减少酸雨产生的措施是A.①②③B.②③④C.①②④D.①③④√解析 煤及其他化石燃料中含S元素,它们燃烧产生的SO2的大量排放是形成酸雨的主要原因。对燃煤产生的尾气进行除硫,少用、不用原煤作燃料,开发清洁能源都可以减少SO2的排放,减少酸雨的产生。燃煤时鼓入足量空气只能提高煤的燃烧效率,不能减少SO2的排放。3.某企业利用下列流程综合处理工厂排放的含有SO2的烟气,以减少其对环境造成的污染。下列有关说法正确的是123A.二氧化硫吸收塔中发生的反应为SO2+SO2-3+H2O===2HSO-3B.氨气吸收塔中发生的反应为HSO-3+OH-===H2O+SO2-3C.上述流程利用的是氧化还原反应原理D.上述流程中SO2可循环利用√A.二氧化硫吸收塔中发生的反应为SO2+SO2-3+H2O===2HSO-3B.氨气吸收塔中发生的反应为HSO-3+OH-===H2O+SO2-3C.上述流程利用的是氧化还原反应原理D.上述流程中SO2可循环利用题述流程中各反应都没有元素化合价的变化,不是氧化还原反应,故C项错误;题述流程吸收SO2,不能循环利用,故D项错误。123返回解析二氧化硫用(NH4)2SO3溶液吸收,发生的离子反应为SO2+SO2-3+H2O===2HSO-3,故A项正确;氨气吸收塔中发生的反应为HSO-3+NH3·H2O===NH+4+SO2-3+H2O,故B项错误;解析二氧化硫用(NH4)2SO3溶液吸收,发生的离子反应为SO2+SO2-3+H2O===2HSO-3,故A项正确;氨气吸收塔中发生的反应为HSO-3+NH3·H2O===NH+4+SO2-3+H2O,故B项错误;



















